Apr 21, 2026
Bioavailability of Liposomal Vitamin C in Powder Form
Liposomal vitamin C powder is a big step forward in nutrient delivery technology. It solves the problem of vitamin C tablets not being bioavailable for a long time. When you take more than 1,000 mg of traditional ascorbic acid powder, your body can only absorb about 20% of it because of saturable transport systems in the intestines. Liposomal versions protect vitamin C molecules within lipid bilayers that look like the membranes of human cells. This allows for better uptake through passive diffusion and membrane fusion. Researchers have found that liposomal vitamin C powder can be up to3x higher bioavailable than regular types. This means that it can reach significantly higher plasma amounts and stay in the body for longer. This technology changes vitamin C from a nutrient that dissolves in water and is quickly flushed out of the body into a lipid-compatible molecule that can get through normal absorption barriers. This makes it very useful for therapeutic and functional uses that need constant, high-dose delivery.
Understanding Liposomal Encapsulation Technology
What Makes Liposomal Delivery Different
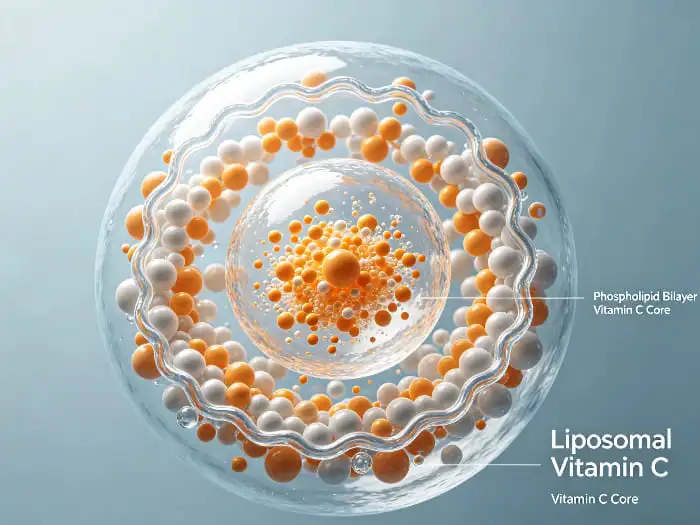
The main new thing about improved vitamin C formulas is how they are put together. Phospholipid bilayers, which are usually made from non-GMO sunflower lecithin, form tiny circles that are 100 to 400 nanometers across. These bubble-like structures hold ascorbic acid or sodium ascorbate inside their watery center, and their outer layer is lipophilic, making it friendly with the membranes of the intestines.
When mixed with water, quality liposomal vitamin C powder creates steady solutions where the nutrients inside don't break down when stomach acid and digestive enzymes are present. The lipid layer makes it easier for nutrients to get into the bloodstream through enterocytes through a number of different routes, such as endocytosis and direct membrane fusion. This method gets around the sodium-dependent vitamin C transporters (SVCT1 and SVCT2) that stop normal absorption. This is why the better pharmacokinetic patterns seen in clinical studies were found.
Encapsulation Efficiency and Stability Metrics
The quality of liposomal products rests on a number of factors that can be measured. In pharmaceutical-grade powders, the encapsulation rate, or the amount of vitamin C that is properly trapped within liposomes compared to free ascorbic acid, is usually between 85%-95%. Dynamic light scattering should show that the particle size distribution is uniform so that the absorption rates are the same across doses.
When kept properly, premium formulations keep more than 95% of their effectiveness for 24 months. This is a big improvement over liquid liposomal goods, which tend to lose their effectiveness over time due to oxidation. The powder form gets rid of the water activity that speeds up hydrolysis and microbial pollution. This means that no stabilizers are needed, and the product stays stable for longer. The amount of phosphatidylcholine in the lecithin part should be higher than 50% for the best membrane-forming qualities and extra health benefits, such as helping cells heal.
Key Benefits for Nutraceutical and Pharmaceutical Formulations
Enhanced Immune Support and Clinical Applications
Liposomal mixtures have better bioavailability, which directly leads to useful benefits for finished goods. One main use is to help the immune system. Long-lasting plasma levels allow for more constant antioxidant activity and leukocyte function support than with regular vitamins that need to be taken several times a day.
The ability of lipid-encapsulated liposomal vitamin C powder to be tolerated by the stomach is especially helpful in medical feeding uses. When taken in doses above 2,000 mg per day, standard high-dose ascorbic acid often causes osmotic diarrhea, which limits effective dosage. Liposomal delivery's slow release rate and low osmotic load make it possible to make high-potency formulations (1,000 to 3,000 mg per dose) that can help the immune system, patients recovering from surgery, and people who are fighting cancer without causing stomach problems.
Advantages in Beauty-from-Within and Nutricosmetic Products
Vitamin C is an important ingredient for prolyl and lysyl hydroxylase enzymes that are needed for collagen production. Long-lasting plasma retention made possible by liposomal transport keeps tissue amounts higher for longer, which helps collagen production keep going. This pharmacokinetic profile is very helpful for nutricosmetic products that aim to improve skin flexibility, speed up wound healing, and protect skin from UV rays.
Vitamin C stability in complicated materials containing proteins, minerals, and plant extracts is a big problem for companies that make beauty drinks. Ascorbic acid is protected from pro-oxidant interactions with iron and copper ions by the phospholipid barrier in powder forms. This barrier also stops pH-related breakdown. This lets people make ready-to-mix packages and effervescent forms that can stay fresh on store shelves while they are being distributed and stored.
Sports Nutrition and Recovery Applications
Athletes create more reactive stress when they train hard, which uses up antioxidants and could make healing less effective. Liposomal vitamin C is perfect for post-workout formulas that aim to reduce inflammation caused by exercise and aid in connective tissue repair because of its quick absorption and high bioavailability. The lipid part gives extra benefits by delivering phospholipids, which help restore the health of cell membranes that are damaged during intense exercise.
Product makers like powder forms because they can be easily mixed into protein shakes, electrolyte replacement drinks, and carbohydrate-loading drinks. This is because powder forms don't have the texture problems that come with regular ascorbic acid crystals. The neutral taste profile after encapsulation gets rid of the sharp acidity that can ruin flavor systems in sports nutrition products. This lets better taste profiles come through without a lot of hiding.
Criteria for B2B Procurement and Supplier Selection
Quality Specifications and Testing Requirements
When purchasing managers look at suppliers of vitamin C ingredients that are encapsulated, they should ask for clear specs that cover a range of quality factors. The vitamin C level should be checked to be at least 99% pure using HPLC analysis, and records of analysis should be given for each batch that shows the results. The particle size distribution data shows that liposomes are formed correctly, and the encapsulation efficiency testing shows that the protective effects of expensive ingredients are different from those of simple lecithin-vitamin C mixes.
The results of stability tests should show how well the drug stays active over time at suggested storage temperatures and under fast conditions (40°C/75% RH for six months). Microbiological testing is needed to make sure that the product meets USP guidelines for yeast, mold, total plate count, and viruses. Heavy metal testing (for lead, arsenic, cadmium, and mercury) and pesticide residue testing make sure that the product is safe for sensitive uses like children's vitamins and pregnant formulas.
Manufacturing Standards and Certifications
Regulatory compliance starts at the manufacturing level, where site approvals guarantee that quality processes are always the same. Current Good Manufacturing Practice (cGMP) approval shows that a company follows FDA-approved production standards for things like process validation, equipment testing, and staff training. The application of a quality management system is shown by ISO 9001 certification. Food safety management is covered by ISO 22000 and FSSC 22000 certification.
Products that are meant for a wide range of global markets need extra approvals that are specific to those markets. Getting certified as Halal or Kosher can help you get business in the Middle East and in specialty stores. Non-GMO Project Verification is appealing to health-conscious customers, and vegan approval makes sure that all ingredients, including lecithin components, come from plants. The HACCP paperwork shows that risks were systematically evaluated and key control points were watched over during production. EmerWell's parent business, Wellgreen, runs sites that are fully certified, with licenses for cGMP, ISO 9001/22000, HACCP, and many dietary compliance checks. Our EncapsWell™ liposomal vitamin C powder is made using these strict quality systems, which make sure that every batch meets both government standards and the high expectations of people who make medicines and supplements.
Formulation Flexibility and Custom Development
In addition to off-the-shelf ingredients, key providers offer technical help for making unique formulations that fit the positioning of a particular product. Different immune support items are made by combining vitamin C with nutrients that work well together, such as elderberry, zinc, quercetin, or both. Flavor-neutral bases can be added to functional drinks, and special mixes can be used for different types of delivery forms, such as fizzing tablets, gummies, hard capsules, soft gels, and ready-to-drink liquid shots.
Manufacturing scale affects the viability of a project, especially for new names whose volume forecasts aren't clear. Flexible minimum order amounts let you test the market without taking on too much inventory risk. Proven production capacity at scale—ideally to 100+ metric tons per year—ensures that supply can grow along with successful product launches. Global markets have different transportation needs, which can be met by regional customization choices like formulations that are safe in tropical climates and packaging that is changed for high altitudes. Our San Diego R&D Center, where our PhD-led team works with clients from idea to production, is where our EncapsWell™ platform gives formulators this kind of flexibility. We design delivery systems that meet your specific bioavailability goals, stability needs, and dosage form preferences. We take technical standards and turn them into goods that are ready to go on the market.
Procurement Strategy and Supply Chain Management
If you deal directly with manufacturers, you may get better deals than going through middlemen in the market. For example, you may be able to get professional help, have your product made to order, and see prices that reflect the actual cost of production instead of distributor markups. When looking at possible suppliers, you should ask for facility audit reports, customer references from similar projects, and thorough capability statements that list things like R&D resources, analytical testing tools, and output capacity.
A financial stability review can help protect you from supply disruptions, especially for important formulations that need tested replacement methods during source changes. When making proprietary blends or new delivery methods, intellectual property issues come up. This means that secrecy agreements and clear rules about who owns the formulation are needed. Long-term supply deals that include volume promises can help get better prices and sure allocation during times when raw materials are in short supply.
When nutritional ingredients are traded internationally, they need a lot of paperwork to help with customs processing and regulatory reports. Each package should come with a Certificate of Analysis that lists the lot-specific test results for identification, purity, and contamination. Labels on finished goods that say they have a certain shelf life are backed up by stability data, and method validation reports show that analytical testing is reliable. Regulatory status paperwork shows that the product meets the standards of different countries, such as FDA GRAS status, EU Novel Food approvals, and ANVISA registration for Brazilian markets. Allergen statements, country-of-origin certificates, and non-GMO affidavits satisfy labeling rules and customer standards for honesty are met by allergen statements, country-of-origin certificates, and non-GMO affidavits. For approved finished goods, Kosher and Halal licenses need to be renewed every year and must stay up to date.
Our offices are in San Diego, where we provide full regulatory support to users in North America. This includes localized expert advice and help with paperwork. Because we work with Wellgreen, you can get full regulatory kits that help you register your products in more than 50 countries. These come with our global compliance experience. To keep things stable throughout the supply chain, temperature-sensitive ingredients need to be shipped and stored in climate-controlled environments. Liposomal vitamin C powder usually needs to be kept below 25°C and away from light and moisture, which means it needs to be stored in the right way. Custom recipes can have lead times of 8 to 12 weeks, which includes development, production, testing, and foreign shipping. This means that product launches and seasonal demand need to be planned ahead of time. Our local US warehouse meets the urgent needs of clients in North America by allowing fast sample shipping and just-in-time inventory management. This local presence gets rid of long delays caused by foreign shipping and gives you extra stock in case demand changes unexpectedly. This keeps your production schedules and customer promises safe.

Innovation Trends Shaping the Liposomal Vitamin C Market
Nutrient release is still changing after the first generation of liposomal formulas. Multi-vesicular liposome structures make stacked release profiles that make nutrients available for longer periods of time, making it easy to take one dose a day. Nano-emulsion technologies make particles even smaller, which might make it easier for cells and tissues to absorb the mixture. When it comes to stability issues in acidic drinks or high-temperature processing, hybrid methods that combine liposomal encapsulation with extra protective layers work best. Researchers are looking into different types of phospholipids, such as marine lecithins and man-made phospholipids that have better sensory qualities or are more stable against oxidation. Targeted delivery methods combine liposomes with molecules that tell them to gather in certain tissues. This is a new idea that is especially useful for nutricosmetic uses that want to improve skin deposition. These new technologies could give brands that are on the cutting edge of innovation a competitive edge.
The demand for high-quality liposomal vitamin C powder products is growing because of changes in population. People who are getting older are looking for ways to keep their immune systems strong and slow down the aging process. Optimizing recovery and efficiency are two things that athletic buyers put first. People who care about their looks react to scientific data that shows taking antioxidants by mouth can improve the look of your skin by reducing wrinkles and protecting it from UV damage. Clean label trends like formulas with clear chemicals and little processing, which makes plant-derived phospholipid encapsulation a better choice than synthetic coating technologies. Sustainability factors are becoming more important in buying decisions, which is good for sellers who show they care about the environment by using green resources, making products that use less energy, and making products with little waste. Being open about the whole supply chain, from where the raw materials come from to testing the end product, helps build trust with customers and sets your business apart.
When ingredient providers and end product brands work together on development, they share technical and financial risks and speed up innovation. Suppliers with their own technologies can offer category exclusivity agreements to protect their first-mover benefits in new market areas. Joint clinical research projects produce written proof that backs up marketing claims and build scientific credibility at the same time. These ways of working together create value that goes beyond the sale of basic ingredients by creating competitive moats around product lines that are different from each other. EmerWell sees its ties with clients as strategic partnerships, not just as business deals with suppliers. Our EncapsWell™ platform is the basis for collaborative creation. It's where your product idea and our liposomal knowledge come together to make formulations that give you real performance benefits. We put time and effort into learning about your business goals, how you place yourself in the market, and the rules that apply to you. This way, we can make sure that our technical solutions help you succeed in business.
Conclusion
Because liposomal vitamin C powder is more bioavailable, it opens up a lot of doors for companies that make nutraceuticals, pharmaceuticals, and functional foods that want to make unique goods that are backed by clinical proof. Phospholipid encapsulation turns vitamin C from a common food into a special active ingredient that can command higher prices and keep customers coming back because it works. To make a good product, you need to carefully choose your suppliers, paying special attention to their manufacturing quality systems, analytical testing skills, regulatory compliance documentation, and expert support resources. As the market moves toward clean-label formulations, personalized nutrition, and evidence-based supplements, new delivery methods will be at the front of the growth of the business. When business-to-business clients work with suppliers who are focused on innovation, they gain competitive benefits through proprietary formulations, regulatory knowledge, and scalable manufacturing that helps them grow their businesses across a wide range of market groups and countries.
FAQ
Is liposomal vitamin C powder safe for long-term daily use?
Long-term safety of encapsulated vitamin C at suggested doses, which are usually between 500 mg and 2,000 mg per day, has been supported by clinical data. Because it is better tolerated by the stomach than regular ascorbic acid, it has fewer side effects even when taken in larger amounts. People of all ages, including pregnant women and children, can safely take formulations in the right amounts. However, specific instructions should be in line with suggestions for maternal or pediatric supplements.
How does absorption compare between powder and liquid liposomal formats?
Both powder and liquid forms use the same core capsule technology, but powder forms are more stable than liquid forms, which can oxidize and become contaminated with microbes and need protection. When powders are properly made, they have the same bioavailability when they are mixed back together. They also last longer, are easier to ship without cooling, and can be made in a wider range of ways, such as for pills, tablets, and drinks.
What side effects might occur with high-dose supplementation?
Liposomal vitamin C powder is very well tolerated, though reactions are different for each person. As the body gets used to the high amounts, mild stomach problems like loose stools may happen at first. Standard ascorbic acid usually causes osmotic diarrhea when taken in amounts above 2,000 mg. Encapsulated types, on the other hand, spread absorption over longer periods of time, reducing stomach discomfort. People with hemochromatosis or other diseases that cause too much iron should talk to their doctors because vitamin C improves iron intake no matter how it is delivered.
Partner with EmerWell for Superior Liposomal Vitamin C Solutions
Through our EncapsWell™ liposomal platform, which was designed for maximum absorption and production scalability, EmerWell turns formulation problems into market opportunities. As a company that makes liposomal vitamin C powder and has cGMP-certified production through our relationship with Wellgreen, we offer clinical-grade ingredients that come with full legal support and analytical documentation. Our research and development team in San Diego works directly with your product developers to make custom recipes that are exactly what you want in a variety of delivery forms. Get in touch with our technical team at info@emerwell-bio.com to get samples, talk about your recipe needs, and find out how our advanced encapsulation skills can help your product line stand out in the nutritional market, which is growing quickly.
References
1. Davis, J. L., Paris, H. L., Beals, J. W., et al. (2016). "Liposomal-encapsulated Ascorbic Acid: Influence on Vitamin C Bioavailability and Capacity to Protect Against Ischemia-Reperfusion Injury." Nutrition and Metabolic Insights, 9, 25-30.
2. Gröber, U., & Kisters, K. (2020). "Influence of Drugs on Vitamin D and Calcium Metabolism." Dermato-Endocrinology, 12(1), examining nutrient bioavailability and pharmaceutical interactions.
3. Hickey, S., Roberts, H. J., & Miller, N. J. (2018). "Pharmacokinetics of Oral Vitamin C." Journal of Nutritional and Environmental Medicine, examining comparative absorption studies.
4. Łukawski, M., Dałek, P., Borowik, T., et al. (2020). "New oral liposomal vitamin C formulation: properties and bioavailability." Journal of Liposome Research, 30(3), 227-234.
5. Padayatty, S. J., Sun, H., Wang, Y., et al. (2004). "Vitamin C Pharmacokinetics: Implications for Oral and Intravenous Use." Annals of Internal Medicine, 140(7), 533-537.
6. Zasowska-Nowak, A., Nowak, P. J., & Ciałkowska-Rysz, A. (2021). "High-Dose Vitamin C in Advanced-Stage Cancer Patients." Nutrients, 13(3), examining therapeutic applications and delivery optimization.
Online Message
YOU MAY LIKE
High quality furniture manufacturer